
Salty diabetes: a case series of hypernatremia presenting with diabetic ketoacidosis
Introduction
Diabetic ketoacidosis (DKA) continues to be the most dreaded complication amongst patients with diabetes mellitus requiring admission to the intensive care unit (ICU). Center of disease control (CDC) reports DKA affecting approximately 30 million patients in the United States and tops the trend in hospitalization affecting population under the age of 45 (1). Due to hyperglycemia-induced osmotic fluid shifts and osmotic diuresis, electrolytes disturbances are expected occurrence in patients with DKA, especially hyponatremia. We detail a case series of two patients who presented with hypernatremia in DKA that would provide insight for clinicians in understanding pathophysiology and treatment.
Case 1
A 28-years-old African American female was transported by emergency medical responders for a worsening mental state. According to the history provided by the family, she awakened on the day of admission complaining of fatigue and sleepiness that progressively worsened. A month prior to the presentation, she developed an episode of diplopia and left leg paresthesia for which she was diagnosed with neuromyelitis optica and received corticosteroids that completed two weeks prior to the presentation of the symptoms. She remained adherent and completed her course of treatment with no further relapse or symptoms. There was no vomiting reported, but she did experience polyuria and suppressed appetite for the last five days. On exam, her Glasgow Coma Scale (GCS) was 10, blood pressure was 128/87 mmHg on left arm, respiratory rate 23 breaths per min, heart rate 133 beats/min, pulse oximetry 98% on ambient air, and temperature 97.2 °F. Clinical examination revealed fruity odor, Kussmaul breathing, and lethargy. CT of the head revealed no evidence of cerebral edema, infarction or abnormal mass. Arterial blood gas (ABG) showed: pH 7.23, pCO2 23 mmHg, pO2 89 mmHg, HCO3 9.6, sodium 175 mmol/L, potassium 3.7 mmol/L, glucose 801 mg/dL, and lactate 1.0 mmol/L. Initial biochemistry analysis showed serum sodium 169 mEq/L, potassium 4.8 mEq/L, chloride 120 mEq/L, bicarbonate 14 mEq/L, albumin 3.7 g/L, and glucose 804 mg/dL. Corrected sodium was calculated to be 186 mEq/L, anion gap 35; delta gap 2.9, and serum osmolality 446 mOsm/kg. Urinalysis showed strongly positive ketone bodies and negative leukocyte esterase and nitrites. Table 1 shows additional biochemical values regarding case 1.
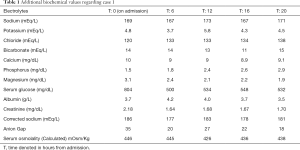
Full table
In emergency department, she received 2 L of 0.9% normal saline that was continued with insulin therapy and electrolytes repletion. Urinalysis had showed no nitrites and leukocyte esterase, but as the patient has used corticosteroids requiring cultures. Urine culture grew Klebsiella pneumonia for which she was started on ceftriaxone. Her serum sodium and serum glucose improved appropriately, but mental status continued with prolonged interval of drowsiness, which was worrisome. CT head showed no intracranial abnormalities that was followed by MRI and MRA brain respectively that were non-conclusive. As the mental status did not improve, lumbar puncture was performed that showed a pattern of viral encephalitis. Cerebrospinal fluid cultures were non-conclusive, but she was commenced on acyclovir. Within 12 h, her mental status improved with responsive to commands and tolerating an oral diet. Patient was continued on acyclovir and transitioned to subcutaneous insulin, who eventually discharged and followed in the clinic with resolution of all the symptoms.
Case 2
A 45-years-old Hispanic female with a history of hypertension, dyslipidemia, DM type 2, and depression presented with nausea and altered sensorium. Two days prior to presentation, the patient started to experience nausea without any episodes of vomiting along with suppressed appetite and decreased oral intake progressing to fatigue, drowsiness and lethargy. Due to change in sensorium, she was not able to take any of her medications, specifically Insulin detemir of 12 units daily. On presentation, she was obtunded with response to painful stimuli and was minimal reception on verbal stimuli. She had a blood pressure of 91/52 mmHg, respiratory rate of 18 breaths per min, heart rate of 89 beats per min, pulse oximetry of 98% on ambient air and temperature of 97.1 °F. On physical exam, she was lethargic, tenderness in periumbilical area on deep palpation and poor skin turgor.
Due to alter sensorium, CT of the head showed no evidence of intracranial abnormalities. A venous blood gas showed pH 7.10, pCO2 18 mmHg, pO2 105 mmHg, HCO3 5.6, sodium 171 mmol/L, potassium 5.6 mmol/L, glucose 685 mg/dL, and lactate 1.3 mmol/L. Initial biochemistry analysis showed serum sodium 168 mEq/L, potassium 6.3 mEq/L, chloride 129 mEq/L, bicarbonate 10 mEq/L, albumin 3.6 g/L, and serum glucose 779 mg/dL. Corrected sodium was calculated to be 179 mEq/L, anion gap 29, delta gap 19, serum osmolality 412 mOsm/kg, alcohol levels <10, and Ketone bodies were strongly positive in the blood and urine. Table 2 shows additional biochemical values regarding case 2.
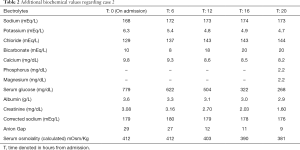
Full table
With working diagnosis of DKA secondary non-adherence to her medications, she received three-1liter bolus of 0.9% normal saline continued with maintenance fluids, and intravenous insulin infusion. Due to high serum osmolarity secondary to hypernatremia and hyperglycemia, patient’s lethargy continued regardless of improvement of serum glucose. Though serum sodium continued to rise, she was maintained on isotonic normal saline and refrained using half-normal saline. Once anion-gap resolved and discontinuing continuous insulin infusion, we switched maintenance fluid to half-normal saline, but patient remained lethargic in a confusional state. Subsequently, patient began to be alert and oriented with intact sensorium that allowed to initiate oral diet and change the choice of maintenance fluid to dextrose 5% water. Serum sodium started to improve and eventually resolved allowing the patient to be discharged on insulin regimen.
Discussion
As uncontrolled hyperglycemia leads to glycosuria and dehydration and acetoacetic acid and Beta-hydroxybutyric acid production ensues due to lack of insulin resulting in anion-gap acidosis contributing to vomiting and further dehydration leading to change in mental status (2). Patients often present with intact and alert mentation but depending on the degree of severity of their acidosis and dehydration they may present or progress to lethargy and/or coma. Once sepsis, cerebral edema or hypoxemia are excluded from consideration for central nervous system (CNS) depression, there are several factors hypothesized to explain, mental status change in DKA. Cerebral hypoperfusion, acidosis, hyperosmolarity and toxic effects of ketone bodies are etiologies considered in the pathogenesis of altered sensorium (2,3). Previous studies attributed pH and osmolality to be the culprit for CNS depression, but recent studies suggest acidotic pH related to the level of consciousness as the main determinant (4-6). In a retrospective study by Nyenwe et al. determining altered sensorium in DKA, acidosis (pH) was the most influential factor with hyperosmolarity playing a synergistic effect and dismissing the role of serum ketone bodies association with altered sensorium (7). Nonetheless acidosis and hyperosmolarity together are considered lethal combination and these subsets of patients present with severe DKA requiring urgent and aggressive treatment due to high mortality rate (7).
Due to resultant osmotic diuresis and hypovolemia in DKA, total body stores of metabolic electrolytes are depleted and not correctly reflected on the laboratory analysis. Hypernatremia may ensue following the loss of water from vomiting, glucose-induced osmotic diuresis and insensible losses, which add to consequential hyperosmolarity. In this case series, we postulate that patients may have had CNS depression secondary to acidosis and concomitant hyperosmolarity, which directly led to impaired ability to ingest water and resulting in acute hypernatremia with DKA. There are numerous pediatric cases reported about hypernatremia in DKA secondary to new-onset Type 1 DM, carbonated carbohydrate beverages and herbal product ingestion, but there are fewer reported cases in adults (8,9). Like carbonated beverages, patient (Case 1) was using corticosteroids that may have played a role in increasing the steady-state of serum glucose to higher levels and compromised to urinary tract infection and encephalitis.
Morbidity and mortality amongst patients who present with acute hypernatremia remain high, and DKA may worsen it further (9). Therefore, choice of intravenous fluids remains crucial for the clinicians to avoid any pitfalls. Treatment should always begin with infusing bolus crystalloid, especially 0.9% normal (isotonic) saline, at a rate of 15–20 milliliters per kilogram per hour or 1–1.5 L during the first hour, to maintain effective plasma osmolality (2,10,11). In Case 2, we continued with choice of normal saline in the maintenance phase as well, because the patient continued to be hypovolemic and dehydrated based on clinical evaluation. Once the patient is hemodynamically stable and hydrated, subsequent use of 0.45% sodium chloride would be possible if the corrected sodium concentration remains above 145 mmol/L. Though one should be cautious about rapid correction of serum glucose and/or serum sodium respectively can precipitate cerebral edema. Therefore, the goal for correction in acute hypernatremia by decreasing plasma sodium concentration at most 2 mol/L/h until plasma concentration is 145 mmol/L (10,11).
From this case series, patients with hypernatremia in DKA presents with clouded consciousness who are significantly dehydrated. With high serum sodium, clinicians may be tempted to initiate treatment with half-normal saline, which they should avoid as these patients require volume-expanders, such as normal (isotonic) saline. Even with appropriate management, patients may continue to be lethargic secondary to high osmolarity that may contribute to prolonged altered sensorium and therefore requiring frequent neurological evaluation. And lastly from our case series and previous pediatric cases, hypernatremia in DKA tends to present in younger-age group in comparison to elderly, which continues to be topic for future research. Therefore, we hope that our case series provide an insight on the pathophysiology and management for the physicians in handling the care of patients with hypernatremia in DKA.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Informed written consent was obtained from the patients for publication of this report.
References
- Benoit SR, Zhang Y, Geiss LS, et al. Trends in Diabetic Ketoacidosis Hospitalizations and In-Hospital Mortality - United States, 2000-2014. MMWR Morb Mortal Wkly Rep 2018;67:362-5. [Crossref] [PubMed]
- Palmer BF, Clegg DJ. Electrolyte and Acid-Base Disturbances in Patients with Diabetes Mellitus. N Engl J Med 2015;373:548-59. [Crossref] [PubMed]
- Gosmanov AR, Gosmanova EO, Kitabchi AE. Hyperglycemic Crises: Diabetic Ketoacidosis (DKA), And Hyperglycemic Hyperosmolar State (HHS). In: Feingold KR, Anawalt B, Boyce A, et al. editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc. 2000.
- Rosival V. The influence of blood hydrogen ion concentration on the level of consciousness in diabetic ketoacidosis. Ann Clin Res 1987;19:23-5. [PubMed]
- Fulop M, Rosenblatt A, Kreitzer SM, et al. Hyperosmolar nature of diabetic coma. Diabetes 1975;24:594-9. [Crossref] [PubMed]
- Edge JA, Roy Y, Bergomi A, et al. Conscious level in children with diabetic ketoacidosis is related to severity of acidosis and not to blood glucose concentration. Pediatr Diabetes 2006;7:11-5. [Crossref] [PubMed]
- Nyenwe EA, Razavi LN, Kitabchi AE, et al. Acidosis: the prime determinant of depressed sensorium in diabetic ketoacidosis. Diabetes Care 2010;33:1837-9. [Crossref] [PubMed]
- Kim HJ, Kim DH, Jun YH, et al. A rare diabetes ketoacidosis in combined severe hypernatremic hyperosmolarity in a new-onset Asian adolescent with type I diabetes. BMJ Case Rep 2014. [Crossref] [PubMed]
- Adrogué HJ, Madias NE. Hypernatremia. N Engl J Med 2000;342:1493-9. [Crossref] [PubMed]
- Kumar V, Nanavati SM, Melki G, et al. A Case of Diabetic Ketoacidosis Presenting with Hypernatremia, Hyperosmolarity, and Altered Sensorium. Case Rep Endocrinol 2018;2018:4806598. [Crossref] [PubMed]
- Sterns RH. Disorders of plasma sodium--causes, consequences, and correction. N Engl J Med 2015;372:55-65. [Crossref] [PubMed]
Cite this article as: Estifan E, Nanavati SM, Kumar V, Gibiezaite S, Michael P. Salty diabetes: a case series of hypernatremia presenting with diabetic ketoacidosis. AME Case Rep 2019;3:27.


