
Vertical rectus abdominis myocutaneous free flap repair of post-pneumonectomy bronchopleural fistula: a case report
Introduction
Bronchopleural fistula (BPF) is an uncommon complication after thoracic surgery yet poses significant morbidity and mortality risk. Treatment of post-surgical BPF varies according to timing of diagnosis and severity, and post-pneumonectomy BPF with empyema portends a particularly challenging problem since both the infection and pleural space must be addressed. The following report describes a patient who underwent right extrapleural pneumonectomy and developed a late BPF with empyema and was successfully treated with open Clagett window (OCW) to address the infection and subsequent staged vertical rectus abdominis myocutaneous (VRAM) free flap repair. We present the following case in accordance with the CARE reporting checklist (available at https://acr.amegroups.com/article/view/10.21037/acr-22-20/rc).
Case presentation
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
A 46-year-old previously healthy male with a prior 12 pack-year smoking history presented with a non-productive cough and was found to have a right upper lobe mass with hilar involvement and pleural implants. Diagnostic right thoracoscopic wedge resection initially demonstrated epithelioid malignant mesothelioma on tissue examination; no other suspicious intra-thoracic lesions were observed during the patient’s index operation. After multi-disciplinary discussion, the patient underwent chemotherapy (cisplatin and pemetrexed for 6 cycles, and bevacizumab as tolerated in setting of hemoptysis) and immunotherapy (ipilimumab and nivolumab) as the disease was initially thought to be unresectable. Despite treatment, his disease progressed to occlude the right mainstem bronchus with right upper lobe collapse and left mainstem bronchus compression. He then underwent urgent palliative right thoracotomy with extrapleural pneumonectomy. The right upper lobe mass was resected to R1 margins but a residual paratracheal tumor was unresectable due to tracheal involvement. The right mainstem bronchus was stapled with a black-load EndoGIA stapler (Medtronic, Minneapolis, MN, USA), and leak test was negative intra-operatively. Additional tissue coverage of the bronchial stump was not performed. The patient received perioperative intravenous steroid therapy due to an ongoing steroid taper in the setting of immunotherapy-induced pneumonitis diagnosed two months before surgery. He was initially started on 80 mg oral prednisone daily which was tapered to 30 mg prior to surgery, and was discharged on 40 mg oral prednisone daily which was tapered over 3 months post-operatively. Final pathology revealed marginal zone B-cell lymphoma without evidence of malignant mesothelioma.
The patient represented on post-operative day (POD) 29 with worsening dyspnea and underwent bronchoscopy in which a left mainstem endobronchial stent was placed for worsening left mainstem bronchus compression. On POD 40 the patient started adjuvant chemotherapy (rituximab, bendamustine) for marginal zone B-cell lymphoma and was also admitted for COVID-19 pneumonia after testing positive with 4 days of reported fevers, dyspnea, and productive cough. He remained hospitalized for 30 days and received high-flow oxygen therapy and courses of remdesivir, prednisone, and tocilizumab while continuing chemotherapy.
On POD 82 the patient underwent bronchoscopy to evaluate the left bronchial stent and the right mainstem bronchus stump was notably completely dehisced with an associated empyema. At that time the patient reported an improving cough with 1.5–3 liters nasal cannula oxygen requirements that were previously attributed to his chemotherapy and COVID-19 sequelae. He was admitted for BPF treatment and underwent right OCW on POD 88 with partial 3-rib resection and washout to address his empyema. A 2 cm BPF was appreciated at the right mainstem bronchus orifice and the pleural cavity was packed with saline-soaked kerlix. The patient underwent daily dressing changes and antibiotic therapy for three months until the pleural cavity was without evidence of infection and white blood cell counts normalized (Figure 1).
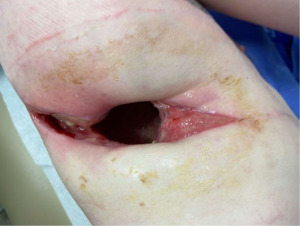
On POD 106 from his OCW, the patient underwent right chest wall reconstruction with VRAM free flap repair to cover the BPF defect and pleural cavity. Partial rib resection was necessary to allow for flap reach to cover the BPF. The entire right rectus abdominis muscle was harvested with preservation of the inferior epigastric vascular pedicle for free flap anastomosis to the ipsilateral thoracodorsal vessels. The flap was inset, rotated obliquely, pressed against the right mainstem bronchial defect to obliterate the BPF, and secured in place with a combination of suture and Tisseal. A skin paddle from the free flap was preserved to close the OCW defect.
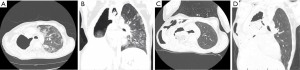
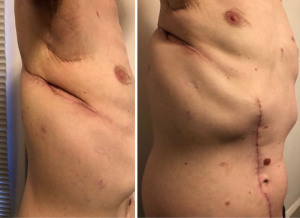
Post-operatively, the patient’s chest tube and Blake drain were removed POD 5 and he received 325 mg aspirin daily until the day of discharge on POD 6. The patient’s only remarkable complication was a persistent subcutaneous emphysema which was confirmed on CT imaging (Figure 2), whereby a right pigtail pleural chest tube was placed on POD 13 remained in place for 39 days until his subcutaneous emphysema completely resolved. The patient’s respiratory status and activity levels improved considerably post-operatively. He still requires low-dose oxygen therapy at home but is able to trial off supplemental oxygen for several hours at a time, which he could not do previously. He has also increased his activities at home and pursues mild-effort activities outside his home. Additionally, he has resumed adjuvant chemotherapy for definitive treatment of his marginal zone lymphoma after recovering successfully from his VRAM free flap reconstruction (Figure 3).
Discussion
The incidence of BPF is up to 20% after pulmonary resection, and occurs more frequently after total pneumonectomy (1,2). Additionally, BPF harbors major morbidity and a mortality risk of up to 50% (3,4). BPF can be classified according to time of diagnosis as early (≤7 days), intermediate (8–30 days), or late (>30 days) (5). Early BPF is usually a technical complication and necessitates urgent surgical intervention. Late BPF is managed according to size and severity and utilizes conservative, endoscopic, and/or surgical modalities (1). Late BPF with infection or empyema requires clearance of infection prior to definitive reconstruction using combined antibiotic therapy with pleural drainage or more definitively, open window thoracostomy such as a Clagett window (4). BPF reconstruction options include muscle (latissimus, pectoralis, serratus, rectus abdominis, intercostal, diaphragm), and soft tissue flaps (omentum, pleura, epicardium) (5). Transverse rectus abdominis myocutaneous flaps have been previously described, but VRAM free flap reconstruction is a more novel technique and has not yet been described in the literature (6).
There are many risk factors for the development of BPF. Patient factors include malnutrition, diabetes, sepsis, infection, immunosuppression, and smoking among other factors. Anatomic factors include right pneumonectomy, a long bronchial stump, neoadjuvant chemoradiation, and tumor positive margins. Other factors include infection (tuberculosis, COVID-19) and steroid use (2,5,7). This patient had significant risk factors for postoperative BPF as he was immunocompromised (due to neoadjuvant and adjuvant chemotherapy, immunotherapy, and perioperative high-dose steroids) and underwent right extrapleural pneumonectomy with tumor positive margins. His COVID-19 pneumonia 6 weeks post-operatively was also a likely negative predictive factor. The influence of COVID-19 infection on development of BPF is unclear, but has been described as a spontaneous complication of COVID-19 pneumonia (7). Nevertheless, tumor shrinkage at the bronchial stump margin via adjuvant chemotherapy likely contributed most this patient’s BPF occurrence.
This patient’s BPF was exceptionally difficult to treat as he had a large right pleural cavity defect from his pneumonectomy and his BPF defect was at the base of the carina. To definitively address his BPF, a reconstruction option that offered a large volume of soft tissue to obliterate the pleural space with sufficient length and flexibility to cover the BPF at the carina was required. Although not reported previously as a reconstructive technique for surgical treatment of BPF, a VRAM free flap utilizes the entire length of rectus abdominis muscle and also may be orientated liberally to conform to large, complex spaces as in this patient’s case. Coordination with plastic surgery for this patient’s VRAM free flap reconstruction was instrumental to optimizing his outcome. Key technical points included harvest of the entire rectus abdominis muscle en-bloc with the overlying skin and soft tissue to also help close the OCW defect, preservation of the inferior epigastric and thoracodorsal vascular pedicles for anastomosis, multiple partial rib resection to create additional length for the flap to reach the carina, and liberal use of suture along the posterior chest wall and Tisseal at the BPF defect to diligently secure the flap in place. In examining post-operative outcomes, there is no evidence to establish the morbidity of VRAM flap reconstruction for BPF defects, but utilization of VRAM flaps for perineal reconstruction has demonstrated somewhat low morbidity with abdominal wall hernia rates observed between 3.3–17% (8,9).
In treating a large late BPF, clearance of infection is critical to reducing mortality. Although OCW requires prolonged wound care, it is the most effective treatment for BPF with associated empyema, with up to 92% successful treatment rates reported in a single institutional case series (4). Once infection is cleared and the pleural cavity can tolerate tissue transfer, reconstruction with soft tissue coverage of the BPF defect may be performed to definitively treat the BPF. The VRAM free flap is a unique repair option for large BPFs as the flap may be orientated freely and provides considerable tissue volume and length to obliterate large pleural defects. VRAM free flaps may also be tolerated with little morbidity and weakness in lieu of remaining abdominal wall musculature (10). Additionally, VRAM flaps can be used in previously irradiated patients where nearby flaps are compromised. However, the long-term effectiveness of VRAM free flap compared to other coverage options in treating BPF are unclear. VRAM free flap reconstruction should be reserved for patients with large BPF defects as less invasive local tissue coverage options may offer similar outcomes with less morbidity in those with small BPF defects, and is relatively contraindicated in patients with compromised inferior epigastric blood supply (prior abdominal wall surgery, ostomy through rectus sheath). Nevertheless, VRAM free flap offers an ideal reconstructive technique to treat post-pneumonectomy BPF and should especially be considered in patients with large tracheobronchial and/or large pleural cavity defects, and those without sufficient local tissue coverage options.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at https://acr.amegroups.com/article/view/10.21037/acr-22-20/rc
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://acr.amegroups.com/article/view/10.21037/acr-22-20/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cusumano G, Alifano M, Lococo F. Endoscopic and surgical treatment for bronchopleural fistula after major lung resection: an enduring challenge. J Thorac Dis 2019;11:S1351-6. [Crossref] [PubMed]
- Zhang C, Pan Y, Zhang RM, et al. Late-onset bronchopleural fistula after lobectomy and adjuvant chemotherapy for lung cancer: A case report and review of the literature. Medicine (Baltimore) 2019;98:e16228. [Crossref] [PubMed]
- Andreetti C, Menna C, D’Andrilli A, et al. Multimodal Treatment for Post-Pneumonectomy Bronchopleural Fistula Associated With Empyema. Ann Thorac Surg 2018;106:e337-9. [Crossref] [PubMed]
- Hysi I, Rousse N, Claret A, et al. Open window thoracostomy and thoracoplasty to manage 90 postpneumonectomy empyemas. Ann Thorac Surg 2011;92:1833-9. [Crossref] [PubMed]
- Dal Agnol G, Vieira A, Oliveira R, et al. Surgical approaches for bronchopleural fistula. Shanghai Chest 2017;1:14. [Crossref]
- Huang JW, Lin YY, Wu NY, et al. Transverse rectus abdominis myocutaneous flap for postpneumonectomy bronchopleural fistula: A case report. Medicine (Baltimore) 2017;96:e6688. [Crossref] [PubMed]
- Habib MB, Mohammad Obeidat I, Ali K, et al. Bronchopleural fistula causing persistent pneumothorax in COVID-19 pneumonia patient with no risk factors. Clin Case Rep 2021;9:e05128. [Crossref] [PubMed]
- Radwan RW, Tang AM, Harries RL, et al. Vertical rectus abdominis flap (VRAM) for perineal reconstruction following pelvic surgery: A systematic review. J Plast Reconstr Aesthet Surg 2021;74:523-9. [Crossref] [PubMed]
- Kim E, Fernando C, McCombie A, et al. Abdominal and perineal hernia rates following vertical rectus abdominis myocutaneous (VRAM) flap reconstruction – a supraregional experience. J Plast Reconstr Aesthet Surg 2022;75:1158-63. [Crossref] [PubMed]
- Jiang L, Jiang GN, He WX, et al. Free rectus abdominis musculocutaneous flap for chronic postoperative empyema. Ann Thorac Surg 2008;85:2147-9. [Crossref] [PubMed]
Cite this article as: Brennan PG, Hsu DS, Banks KC, Maxim CL, Hornik B, Velotta JB. Vertical rectus abdominis myocutaneous free flap repair of post-pneumonectomy bronchopleural fistula: a case report. AME Case Rep 2022;6:33.


