A case report of coronary pulmonary artery fistula detected by transthoracic echocardiography in an elderly patient with dyspnea
Introduction
Coronary pulmonary artery fistula (CPAF) is a rare entity with a prevalence of 0.32–0.68% in the population and presents with multiple clinical settings (1-4). It could be either congenital or acquired (1), and therefore is seen in both the neonatal and elderly patients. Although chest pain and shortness of breath are the most common complains, 31% of CPAF cases are asymptomatic (1). Whereas CPAFs mostly are diagnosed by invasive coronary angiography (ICA) or coronary computed tomography angiography (CCTA), a few of them are identified by transthoracic echocardiography (TTE) (5). We report a case of suspected congenital CPAF accidentally detected by TTE in a 93-year-old female who presented with acute respiratory distress on the setting of community-acquired pneumonia, diastolic heart failure, ischemic heart disease, pulmonary hypertension, chronic kidney disease, and hypertension. Although there is one previously reported 83-year-old patient with congenital CPAF (6), it is very uncommon for a patient to have the same entity detected at the age of 93 especially by TTE.
We present the following case in accordance with the CARE Reporting checklist (available at
Case presentation
A 93-year-old female was admitted to a tertiary hospital for orthopnea, fever, bilateral pleuritic chest pain, and productive cough with yellowish sputum for 7 days. Vital signs showed a heart rate of 102/minute, arterial blood pressure of 150/90 mmHg, temperature of 39 °C, respiratory rate of 22/minute, and saturated pulse oximetry of 87% with room air. She was alert and oriented. Her cardiac examination showed gallop S3, regular rhythm, normal S1 and S2 with no murmur. Her chest examination disclosed scattered crackles. Other abnormal physical findings included conjunctival pallor and bilateral ankle edema. Her past medical history included 20-year hypertension, chronic kidney disease, and no chest trauma or surgery.
The chest radiograph demonstrated right-sided lung consolidate infiltration, bilateral hydrothorax, and no enlarged cardiac silhouette. T wave inversion on V1-V4 with no ST changes were seen on the electrocardiogram (ECG). Her serum lab results: WBC 17.4 G/L (neutrophil 87.3%), procalcitonin 44.67 ng/mL, BNP 974.7 pg/mL, creatinine 2.42 mg/dL (estimated glomerular filtration rate of 21 ml/min/1.73 m2). The thoracocentesis analysis was consistent with the transudate fluid.
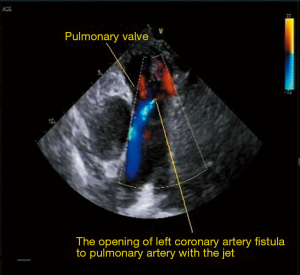
The TTE disclosed: (I) mild concentric left ventricular hypertrophy with left ventricular ejection fraction =65% and E<A; (II) moderate left atrial enlargement (41 mm) (Video 1); (III) mild ascending aortic enlargement (d=42 mm) (Video 1); (IV) right and left coronary artery originating from the right and left Valsalva sinus respectively; (V) left coronary artery tortuously enlarges (d=9 mm) (Figure 1) and drains into the pulmonary arterial trunk right above the pulmonary valve (Figure 2; Videos 2,3), mild pulmonary hypertension with PAP =45 mmHg, mild pulmonary regurgitation; (VI) mild mitral regurgitation, mild aortic regurgitation (Video 4); (VII) mild inferior wall hypokinesis.
The patient was diagnosed with acute respiratory distress, community-acquired pneumonia, diastolic heart failure complicated with bilateral hydrothorax, CPAF, mild pulmonary hypertension, ischemic heart disease, chronic kidney disease, and hypertension. The treatment regimen included loop diuretic (40 mg of intravenous furosemide), oxygen therapy, antibiotic (750 mg of intravenous levofloxacin), antiplatelet (75 mg of oral clopidogrel), and statin (40 mg of oral atorvastatin) from January 11 to January 23, 2017. She recovered and was discharged after 13 days. The regimen at home included furosemide (40 mg orally), clopidogrel (75 mg orally), and atorvastatin (40 mg orally). However, she refused the follow-up. A CCTA or ICA was not performed due to her poor renal function.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient.
Discussion
Coronary artery fistula (CAF) is a rare entity in the population. Historical ICA detected the CAF prevalence of 0.05–0.25% with the right coronary artery and right heart chamber as the most often involved sites (1,7-9). However multiple recent studies using CCTA revealed the higher CAF prevalence (0.88%) with the most common drainage site is the main pulmonary artery (1,2). The prevalence of CPAF ranges from 0.32% to 0.68% (1-4). Most of them are between the left coronary arterial system and the main pulmonary artery which is also found in our case (1).
ICA is not considered gold standard for the coronary artery anomaly as CCTA diagnosed 20% and 80.9% cases of CPAF missed by ICA and TTE respectively (5). The ICA may under-detect the fistula due to the dilution of contrast from a very small artery calibers into the low-pressured pulmonary artery, the steal phenomenon in distal arterial segments, and the limitation of two-dimensional angiographic angle (1,5). The CCTA visualizes not only the abnormal blush of contrast in the relatively less opacified pulmonary trunk but also the fistulous connection in a three-dimensional imaging reconstruction (1,5). Meanwhile TTE has the lowest probability in detecting only 19.1% cases of CPAF due to poor acoustic transmissibility, two-dimensional imaging style, lower spatial resolution, and strong subjective judgment (5). Our CPAF case was diagnosed by a clear visualization of TTE, however could not be confirmed by CCTA or ICA due to the patient’s poor renal function.
Patients with CPAF present with various clinical settings. Although most of the CPAFs are hypothesized resulting from the abnormal persistence of the pulmonary sinus anlage which connects with the aortic sinus anlage during the embryological process (1,10); it may be acquired after the procedures such as coronary artery bypass grafting, valve replacement, endomyocardial biopsy, or chest trauma (1). As a result, CPAFs are seen from the neonatal to the elderly population. A review of 103 patients with CPAF showed the ages range from newborn to 88 years (mean =46.1 years) and 63% were male (1). Although chest pain, dyspnea, and murmur are seen in 39%, 25%, and 37% of CPAF cases respectively; 31% present with neither symptoms nor signs (1). Our patient has had no cardiac intervention or chest trauma, and therefore, to our knowledge, may be the oldest case report with congenital CPAF. Mild pulmonary hypertension is not uncommon in the elderly, but in our case, it may result from the small left-to-right shunt because we could not find out any other causes during her hospitalization. Despite no angina, her myocardial ischemic sign on the ECG and TTE may be a consequence of the coronary steal phenomenon which is frequently seen in the coronary artery fistulas. The coronary steal phenomenon is a condition in which the low pressure sink of the fistula allows preferential flow to get through it and can lead to low perfusion in the distal native coronary artery (11). However, aging-related atherosclerosis remains a possible cause of myocardial ischemia as the fistula is small and the patient is in advanced age.
Although the potential complications of CPAF include myocardial ischemia, pulmonary hypertension, congestive heart failure, and coronary aneurysm; there is no current guideline for the treatment of CPAF (1). In spite of its efficiency and safety, it is still controversial if the surgical ligation or transcatheter procedure using embolism coils should be performed to close the fistula in asymptomatic patients (1,12-14). In addition, the spontaneous closure of CPAF was also reported (15). Therefore, the interventional decision should be based on multiple factors including symptoms; the size, type, and hemodynamic effects of CPAF; as well as concomitant cardiovascular diseases (1,16). However bacterial endocarditis prophylaxis is recommended in all patients with CPAF (1,7). Our patient is in advanced age and with multiple comorbidities, therefore should be treated by the conservative medical therapy with diuretics.
Conclusions
CPAF is a rare entity which can be congenital or acquired. Besides CCTA and ICA, TTE is capable of detecting CPAF. The patient may present with multiple clinical settings. The treatment decision therefore should be individualized.
Acknowledgments
We are grateful for the assistance given by Dr. Vo Thi Phuong Loan (cardiologist, Cho Ray Hospital) and Dr. Nguyen Thanh Huan (cardiologist, University of Medicine and Pharmacy at Ho Chi Minh City).
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting Checklist. Available at http://dx.doi.org/10.21037/acr-20-100
Peer Review File: Available at http://dx.doi.org/10.21037/acr-20-100
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/acr-20-100). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for the review by the Editor-in-Chief of this journal. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Verdini D, Vargas D, Kuo A, et al. Coronary-Pulmonary Artery Fistulas: A Systematic Review. J Thorac Imaging 2016;31:380-90. [Crossref] [PubMed]
- Lim JJ, Jung JI, Lee BY, et al. Prevalence and types of coronary artery fistulas detected with coronary CT angiography. AJR Am J Roentgenol 2014;203:W237-43. [Crossref] [PubMed]
- Kim MS, Jung JI, Chun HJ. Coronary to pulmonary artery fistula: morphologic features at multidetector CT. Int J Cardiovasc Imaging 2010;26:273-80. [Crossref] [PubMed]
- Yun H, Zeng MS, Yang S, et al. Congenital coronary artery fistulas: dual-source CT findings from consecutive 6,624 patients with suspected or confirmed coronary artery disease. Chin Med J (Engl) 2011;124:4172-7. [PubMed]
- Li JL, Huang L, Zhu W, et al. The evaluation of coronary artery-to-pulmonary artery fistula in adulthood on 256-slice CT coronary angiography: Comparison with coronary catheter angiography and transthoracic echocardiography. J Cardiovasc Comput Tomogr 2019;13:75-80. [Crossref] [PubMed]
- Tachibana M, Mukouhara N, Hirami R, et al. Double congenital fistulae with aneurysm diagnosed by combining imaging modalities. Acta Med Okayama 2013;67:305-9. [PubMed]
- Zenooz NA, Habibi R, Mammen L, et al. Coronary artery fistulas: CT findings. Radiographics 2009;29:781-9. [Crossref] [PubMed]
- Lowe JE, Oldham HN Jr, Sabiston DC Jr. Surgical management of congenital coronary artery fistulas. Ann Surg 1981;194:373-80. [Crossref] [PubMed]
- Levin DC, Fellows KE, Abrams HL. Hemodynamically significant primary anomalies of the coronary arteries. Angiographic aspects. Circulation 1978;58:25-34. [Crossref] [PubMed]
- Vaidyanathan KR, Theodore SA, Sankar MN, et al. Coronary artery to pulmonary artery fistula with dual origin--embryological, clinical and surgical significance. Eur J Cardiothorac Surg 2007;31:318-9. [Crossref] [PubMed]
- Reddy G, Davies JE, Holmes DR, et al. Coronary artery fistulae. Circ Cardiovasc Interv 2015;8:e003062. [Crossref] [PubMed]
- El-Sabawi B, Al-Hijji MA, Eleid MF, et al. Transcatheter closure of coronary artery fistula: A 21-year experience. Catheter Cardiovasc Interv 2020;96:311-9. [Crossref] [PubMed]
- Shah AH, Osten M, Benson L, et al. Long-term outcomes of percutaneous closure of coronary artery fistulae in the adult: A single-center experience. Catheter Cardiovasc Interv 2020;95:939-48. [Crossref] [PubMed]
- Kamiya H, Yasuda T, Nagamine H, et al. Surgical treatment of congenital coronary artery fistulas: 27 years' experience and a review of the literature. J Card Surg 2002;17:173-7. [Crossref] [PubMed]
- Iwaki T, Shimizu M, Mabuchi H. Images in cardiology. A case of spontaneous closure of coronary artery fistula with familial hypercholesterolaemia. Heart 2002;87:219. [Crossref] [PubMed]
- Oh JH, Lee HW, Cha KS. Hemodynamic significance of coronary cameral fistula assessed by fractional flow reserve. Korean Circ J 2012;42:845-8. [Crossref] [PubMed]
Cite this article as: Truong T, Nguyen HTT, Phan VTX, Ly MHP, Phan VTT, Phan TA, Phan HH, Tran P. A case report of coronary pulmonary artery fistula detected by transthoracic echocardiography in an elderly patient with dyspnea. AME Case Rep 2021;5:10.