Use of custom made 3-dimensional printed surgical guide for manubrio-sternal resection of solitary breast cancer metastasis: case report
Introduction
Chest wall resection for malignant tumours can be challenging, particularly when the lesion is deep within the bony structure of the chest. Achieving adequate soft tissue and bone margins would optimize oncological outcomes, however, accurate intra-operative identification of these boundaries can be very difficult. We describe a case of a 44-year-old lady with manubrio-sternal metastasis from breast cancer who underwent precision chest wall resection using pre-procedural imaging designed 3-dimensional (3D) printed surgical guides to rapidly define soft tissue and deep bone resection boundaries. We present the following case in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/acr.2020.03.08).
Case presentation
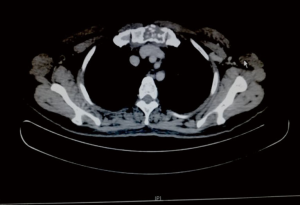
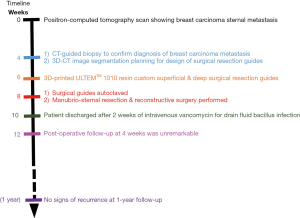
A 44-year-old lady was diagnosed to have invasive ductal carcinoma of the left breast in October 2016. She underwent left modified radical mastectomy in early 2017, with pathology revealing no residual tumor and negative for any lymph node involvement. She was started on hormonal therapy. A year later, she had sternal pain, and a 4.4-cm solitary metastasis to the manubrium was confirmed by positron emission tomography-computed tomography (PET-CT) scan (Figure 1) and CT-guided biopsy. Multi-disciplinary decision was for resection of the sternal metastasis and chest wall reconstruction.
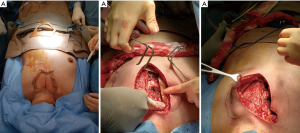
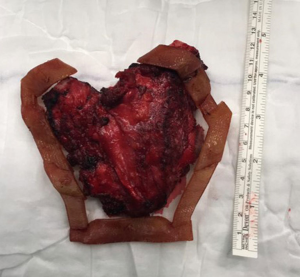
Pre-operative CT scan was analyzed with Mimics Version 19 (Materialise, Neuven, Belgium) to segment out the tumour with a 2-cm resection margin on the bone, and marked on the reconstructed 3D computer model. A superficial surgical guide for skin incision (Figure 2) and deep surgical guide for bone margin (Figure 3) were designed by Persona Surgical Modelling Co., Ltd. (Hong Kong, China). The surgical guides were printed with ULTEMTM 1010 resin (Stratasys, Minnesota, USA) which is an fused deposition modeling (FDM) thermoplastic which offers excellent strength, thermal stability and ability to withstand sterilization by steam autoclaving. During the surgery, the 3D-printed personalised superficial and deep surgical guides were used to determine the pre-determined boundaries of resection. The superficial skin incision surgical guide was used to mark the skin to determine the extent of soft tissue resection superficial to the sternal metastases (Figure 4A). After obtaining adequate exposure of the manubrium and corresponding ribs and clavicles, the deep surgical guide that was designed to have 2-cm resection margins around the tumour was placed directly onto the chest wall (Figure 4B,C). The manubrium, clavicular head, insertion of the first and second rib as well as the upper sternum just above the third rib were resected en-bloc according to the 3D-printed surgical guide (Figure 5). The chest wall was then reconstructed using PermacolTM (Medtronic Inc., USA) patch to cover the deep defect, and MatrixRIBTM titanium rib (DePuy Synthes, Johnson and Johnson, USA) was implanted to reconnect the 1st ribs, similar to approach previously described (1). The clavicles were approximated with sternal wires. A negative pressure drain was placed afterwards. Post-operatively, the patient’s recovery was mildly complicated with positive culture of Bacillus species from drain fluid requiring 2 weeks of intravenous vancomycin treatment. The patient remained free from local recurrence on CT scan 1-year after resection (Figure 6).

Discussion
The utility of 3D printing in surgery have come a long way in terms of its reliability and efficacy. Creating a 3D-reconstructed image or printed model of bone structures extracted from CT scan data allows not only for precise prosthesis design but also provides adequate visualization of the defect for preoperative surgical evaluation and thorough planning of the repair (2). In doing so, intraoperative time could be lessened, morbidity and reoperation could be minimized and better outcomes achieved.
In this case of sternal metastases, we adopted a novel approach and concept in chest wall resection of pre-operatively planning for the surgery using 3D image segmentation, design an ideal soft tissue incision perimeter and bone resection boundary to ensure desirable oncological margins, and subsequently print out superficial and deep surgical guides that allow the surgeon to execute the precision resection. By being more accurate and certain of the resection boundaries and thus better able to predict the resulting chest wall defect, the surgical team can more effectively plan for the subsequent reconstruction of the chest wall defect and materials needed. Understanding the size and characteristics of the defect prior to surgery could be important for successful outcome in chest wall reconstruction especially when using the MatrixRib system (1). Furthermore, as pre-fabricated patient-specific biomechanically compatible 3D-printed implants for chest wall reconstruction becomes more popular, accurate resection of the chest wall becomes paramount (3).
One of the challenges of our approach is the ability to 3D print the surgical guides in reasonable time and use a material that is non-toxic and could be placed on the patient in a sterile operating environment. ULTEMTM 1010 resin is one of the strongest FDM thermoplastics, with high heat resistance and thermal stability up to 216 degrees Celsius. It is able to withstand autoclave operations associated with sterilization. Furthermore, the certified grade form of this plastic is biocompatible and approved for food contact with United States Plastic (USP) Corp NSF51 and ISO10993/USP Class VI certifications. The resin’s strength is also important as a surgical guide that easily fragments or chips would not only make the guide useless, but also create a nightmare scenario of foreign body retrieval from surgical site.
Our report showed the feasibility and safety of using pre-planned 3D-printed surgical guides to help define incision and resection borders during surgery, but is limited by the single report only, thus may not be generalizable to other locations in the chest or other clinical scenarios. Furthermore, we do not have the long-term follow-up or comparative data on whether this method saves time and improves the resection margin. Larger prospective studies are needed to answer these questions.
Conclusions
In conclusion, the application of 3D printing is ever increasing in various fields of surgery. Its role has been well defined in surgical planning, education, custom made implants and stents, and even as templates for guiding lung nodule localization (4). Our case has demonstrated an additional role for 3D printing in surgery of creating surgical resection guides that provides the surgeon with precise and rapid real-time on-table “road-map” for desirable tumour resection margins. The potential benefits of using such guides will require further prospective studies.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at http://dx.doi.org/10.21037/acr.2020.03.08
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/acr.2020.03.08). CSHN reports personal fees from Johnson & Johnson, personal fees from Medtronic, outside the submitted work. CSHN serves as an unpaid editorial board member of AME Case Reports from Jun 2016 to May 2021. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the noncommercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
sReferences
- Ng CS, Ho AM, Lau RW, et al. Chest wall reconstruction with MatrixRib system: avoiding pitfalls. Interact Cardiovasc Thorac Surg 2014;18:402-3. [Crossref] [PubMed]
- Wen X, Gao S, Feng J, et al. Chest-wall reconstruction with a customized titanium-alloy prosthesis fabricated by 3D printing and rapid prototyping. J Cardiothorac Surg 2018;13:4. [Crossref] [PubMed]
- Moradiellos J, Amor S, Córdoba M, et al. Functional Chest Wall Reconstruction With a Biomechanical Three-Dimensionally Printed Implant. Ann Thorac Surg 2017;103:e389-91. [Crossref] [PubMed]
- Kwok JKS, Lau RWH, Zhao ZR, et al. Multi-dimensional printing in thoracic surgery: current and future applications. J Thorac Dis 2018;10:S756-63. [Crossref] [PubMed]
Cite this article as: Fernandez RAS, Lau RWH, Yu PSY, Siu ICH, Chan JWY, Ng CSH. Use of custom made 3-dimensional printed surgical guide for manubrio-sternal resection of solitary breast cancer metastasis: case report. AME Case Rep 2020;4:12.