
Successful revascularization of right coronary artery chronic total occlusion by the antegrade and retrograde approaches
Introduction
Chronic total occlusion (CTO) recanalization of coronary arteries remains one of the most challenging procedures in interventional cardiology, despite the availability of new specialized guidewires, microcatheters, and dedicated devices, as well as many sophisticated techniques for crossing occluded arteries and the widespread use of drug-eluting stents (DES) (1,2). Successful CTO percutaneous coronary intervention (PCI) has been associated with better long-term outcomes and an improvement in mortality, left ventricular function and quality of life (3). In addition, the introduction of the retrograde approach, especially “the Hybrid algorithm” and the consensus document from the CTO Club, have significantly and impressively increased CTO PCI adoption, procedural efficiency, and success rate (4). The hybrid algorithm was initially reported in 2012, and the algorithm is predicated on four angiographic features seen on dual catheter angiography: (I) anatomy of the lesion’s proximal and distal caps, (II) anatomy of the vessel distal to the lesion’s distal cap, (III) the presence of useable collaterals, and (IV) the length of the lesion occlusion segment. These characteristics inform the operator whether to start with an antegrade or retrograde approach (5). In the present report, we describe a patient with multivessel disease who underwent total revascularization by staged PCI. Here, we report the combined interventional procedural steps to successful revascularization of the RCA CTO under the guideline of the hybrid algorithm.
Case presentation
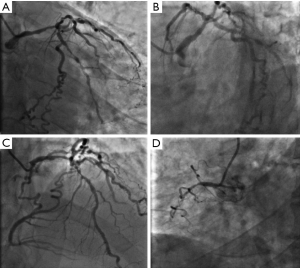
A 58-year-old man presenting with symptoms of effort angina was admitted to our hospital. He was diagnosed with silent myocardial ischemia that was detected by myocardial perfusion scintigraphy. The patient had some coronary risk factors, including current smoking, hyperlipidemia, and hypertension, which required optimal medical treatment. An electrocardiogram suggested deep Q wave inversion in leads II, III and AVF, and an echocardiographic examination showed a normal left ventricular ejection fraction and hypokinesis in the inferolateral wall during hospitalization. Blood tests showed no abnormal result apart from an elevated triglyceride level (5.79 g/L). He underwent coronary angiography upon admission (Figure 1), which indicated severe stenosis of three vessels: the proximal right coronary artery (RCA) had CTO, the proximal segment of the left anterior descending (LAD) artery had 80% stenosis and the proximal segment of the left circumflex artery (LCX) had 90% stenosis with calcification. The SYNTAX score was higher than 33. Coronary artery bypass graft surgery (CABG) was initially recommended; however, he refused CABG and obtained written informed consent. Consequently, we planned a staged PCI, and endovascular treatment to the left coronary artery was recommended as a first strategy. Expecting the development of collateral circulation and improvement of the patient’s tolerance, RCA CTO was tried 3 months after these procedures.
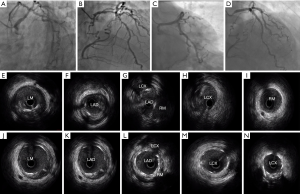
The procedure of left coronary artery PCI was shown in Figure 2. First, Medina 1,1,1 coronary bifurcation lesions in the left main artery (Figure 2A,B) were assessed with intravascular ultrasound (IVUS) (Figure 2E,F,G,H,I). Revascularization of the left coronary artery was successfully implemented by systematic two-stent strategies (culotte technique): 2.75×38 and 3.0×38 mm EXCEL stents (Biosensor/Jiwei Co., Shandong, China) were implanted into the LM-LAD artery; and 2.5×28 and 3.0×16 mm EXCEL stents (Biosensor/Jiwei Co., Shandong, China) were implanted into the LM-LCX artery. Final angiography (Figure 2C,D) and IVUS (Figure 2J,K,L,M,N) showed an optimal result with flow competition in the RCA posterior descending branch (PD) and the posterior left ventricle branch (PL).
After three months, this patient’s re-angiogram in our ward showed that the left coronary artery stents were patent (Figure 3A,B,C) but that proximal stenosis of the RCA had worsened to CTO with several lateral bridge branches in the ostial at an estimated length of 60 mm. The approach locations were the bilateral sites (Figure 3D); an antegrade guiding catheter, AL 0.75 6 Fr (Terumo Corp, Tokyo, Japan), was advanced into the ostium of the RCA via the right radial artery, and a retrograde guiding catheter, EBU 3.5 7 Fr, was advanced via the right femoral artery. Intravenous heparin was administered to maintain an activated clotting (ACT) time of >350 s. Bilateral coronary angiography showed proximal cap ambiguity, the distal vessel ending in bifurcation, and the collateral artery from the LCX having good epicardial collaterals and poor septal branches from LAD. The J-CTO score was 3. According to the hybrid algorithm, there is a main question that determines whether the primary approach is antegrade or retrograde. The procedure was started with the antegrade approach because this was the first attempt, and the microchannel in the CTO lesions could be found. If it had failed, the retrograde approach was planned as the next step.
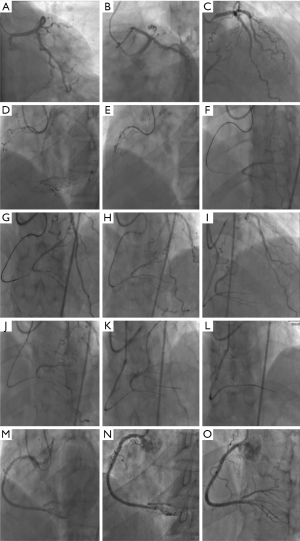
First, a tapered guidewire Fielder XT-R (Asahi, Intecc, Co., Ltd., Japan) and a microcatheter (Corsair 135 cm, Asahi Intecc) were used. We managed to insert the wire into the proximal cap carefully and gradually advanced it to the body of the CTO. Dual injection maintained the wire in the direction of the vessel to avoid straying into the branch. However, the wire tip could not be advanced continually. It followed the microcatheter into the CTO body, and then the wire was exchanged to a Fielder XT-A and Gaia First (Asahi, Intecc). Regardless of the parallel wire technique with double lumen catheter Crusade (Kaneka, Osaka, Japan), the wires went into the distal vessel of branch, not to the main vessel, but this a false lumen. Finally, the Gaia Second guidewire was used to puncture the distal cap and subsequently cross the CTO segment into the distal PD branch. The attempt to adjust the guidewire into the PL was not successful due to the extreme angulation of the distal RCA-PL. Worrying that repeated wire puncture into the distal cap would lead to subintimal hematoma and affect the patency of PL or PD, we finished the antegrade preparation and moved to the retrograde approach (Figure 3E,F,G,H,I).
The guidewire Sion (Asahi Intecc) and Finecross 150 cm microcatheter (MC) were passed through the epicardial collateral via a previous stent strut. To track the Finecross, we dilated the stent strut with a 2.0/15 mm semi-compliant balloon (TERK; St. Jude Medical, Little Canada, MN, USA). Because the injection of isosorbide dinitrate via the MC tip dramatically visualized the channel to the PL, Sion and MC reached the CTO exit. Exchange of the Fielder XT-R and the Gaia First and Gaia Second guidewires all failed to successfully traverse the CTO lesions due to severe angulation at the CTO exit site. We decided to leave the retrograde guidewire in the distal vascular branch and managed to insert the antegrade wire by entering the same vascular cavity as the retrograde guidewire (Figure 3J,K,L). Next, the MC was advanced into the distal branch and demonstrated the true lumen position by blood aspiration. Three DES (Xience Prime 2.5×38, 3.0×38 and 3.5×23 mm, Abbott Vascular, Chicago, USA) were deployed from the distal to ostial RCA. After postdilatation by a Quantum Mavericks 3.0×20 mm (Medtronic, Dublin, Ireland) noncompliant balloon catheter (Figure 3M,N,O), the antegrade final angiogram was optimal, and the epicardial collaterals were not damaged. The entire PCI procedure is presented as a schema in the Figures 3,S1.

Discussion
In this article, we present a patient with multivessel disease, and CABG is considered the standard treatment (6). However, physicians frequently experience a dilemma in recommending CABG to patients with multivessel disease who refuse and insist on undergoing PCI. Patient preference is one of the most important factors to consider in daily clinical practice because it may impact the final decision of the revascularization strategy and influence the relatively lower performance of CABG. Thus, complete revascularization or staged PCI is very important. Theoretically, total revascularization of lesions may reduce the ischemic burden and the incidence of ischemic events and obviate the need for repeat procedures. In contrast, it may cause coronary reserve reduction and a higher risk of renal failure from prolonged procedures and additional contrast use. This patient had multivessel disease and RCA-CTO. Intervention in the left coronary artery was associated with reduction of the symptoms of angina and an increase in the endurance for the next operation.
Due to the recent widespread use of coronary intervention technology in clinics, the coronary intermediate lesion detection rate has increased significantly. CTO lesions occur in 10–20% of patients. With the development of a systematic algorithm for CTO PCI, the success rate of CTO PCI has also clearly improved (7,8). Patients in whom CTO PCI was successful had better outcomes compared with those in whom CTO PCI failed (9). Furthermore, procedural failure was due to the inability to cross the lesion with a guidewire in 70% of cases, or less commonly, in 30% of cases (10), due to a complication. Only a few cases of successful direct antegrade wire crossing CTO lesions have been reported in practice. Therefore, the combination of antegrade and retrograde approaches can help interventionists overcome problems encountered in CTO PCI.
In the present patient, although the segment of RCA occlusion was long and the proximal cap was ambiguous, the “antegrade preparation first” philosophy was prompted (2). The parallel wire technique and wire escalation is the basic technique of the antegrade approach. A sufficient antegrade approach attempt can reduce the time that the retrograde system is engaged in the donor artery, in addition to the CTO territory ischemic time, and donor artery risks. However, the antegrade attempt should be stopped when an angiographic image of the hematoma is observed, and a large subintimal space should be avoided. The selection of the first guide wire in an antegrade fashion is critical to find the micro channel or loose tissue and increase the probability of entering the true lumen (11). Once the soft guide wire, such as Fielder XT-r or Gaia First (Asahi Intecc), can be advanced into the proximal cap of CTO, it is likely to be located in the true lumen or intravascular structure. In our case, we chose the Fielder Xt-r guide wire with a Corsair backup to push into the CTO body. It was difficult for wire advancement to be continued, and the wire was exchanged to Gaia Second (Asahi, Intecc). All attempts to pass through the lesions into the true lumen of RCA-PL failed. A timely shift to perform the retrograde technique was completed to keep the PL open. Naturally, the ambiguity of the distal cap should be overcome with IVUS guidance. However, the IVUS catheter could not be placed in the distal RCA CTO site, making it impossible to locate the exact origin of the distal cap on IVUS. Finally, 3 DESs were successfully deployed from the distal to ostial RCA. The final result was optimal, and the patient suffered no remarkable events during hospitalization.
Conclusions
In summary, under the guideline of a hybrid algorithm and using the staged procedures, we were successful in interventional revascularization of a multivessel disease complicated with RCA CTO lesions. The combination of the antegrade and retrograde approaches can enable a safe and effective recanalization of chronically occluded vessels with complex lesions and enhance the ability to successfully cross a CTO with all available CTO crossing strategies.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Harding SA, Wu EB, Lo S, et al. A New Algorithm for Crossing Chronic Total Occlusions From the Asia Pacific Chronic Total Occlusion Club. JACC Cardiovasc Interv 2017;10:2135-43. [Crossref] [PubMed]
- Riley RF, Walsh SJ, Kirtane AJ, et al. Algorithmic solutions to common problems encountered during chronic total occlusion angioplasty: The algorithms within the algorithm. Catheter Cardiovasc Interv 2019;93:286-97. [Crossref] [PubMed]
- Lee SW, Lee PH, Ahn JM, et al. Randomized Trial Evaluating Percutaneous Coronary Intervention for the Treatment of Chronic Total Occlusion: The DECISION-CTO Trial. Circulation 2019;139:1674-83. [Crossref] [PubMed]
- Hatem R, Finn MT, Riley RF, et al. Zero contrast retrograde chronic total occlusions percutaneous coronary intervention: a case series. Eur Heart J Case Rep 2018;2:1-5. [PubMed]
- Galassi AR, Sianos G, Werner GS, et al. Retrograde Recanalization of Chronic Total Occlusions in Europe: Procedural, In-Hospital, and Long-Term Outcomes From the Multicenter ERCTO Registry. J Am Coll Cardiol 2015;65:2388-400. [Crossref] [PubMed]
- Escaned J, Collet C, Ryan N, et al. Clinical outcomes of state-of-the-art percutaneous coronary revascularization in patients with de novo three vessel disease: 1-year results of the SYNTAX II study. Eur Heart J 2017;38:3124-34. [Crossref] [PubMed]
- Choi JH, Chang SA, Choi JO, et al. Frequency of myocardial infarction and its relationship to angiographic collateral flow in territories supplied by chronically occluded coronary arteries. Circulation 2013;127:703-9. [Crossref] [PubMed]
- Nicholson W, Harvey J, Dhawan R. E-CART (ElectroCautery-Assisted Re-enTry) of an Aorto-Ostial Right Coronary Artery Chronic Total Occlusion: First-in-Man. JACC Cardiovasc Interv 2016;9:2356-8. [Crossref] [PubMed]
- Li R, Yang S, Tang L, et al. Meta-analysis of the effect of percutaneous coronary intervention on chronic total coronary occlusions. J Cardiothorac Surg 2014;9:41. [Crossref] [PubMed]
- Toma A, Stähli BE, Gick M, et al. Impact of multi-vessel versus single-vessel disease on outcomes after percutaneous coronary interventions for chronic total occlusions. Clin Res Cardiol 2017;106:428-35. [Crossref] [PubMed]
- Joyal D, Thompson CA, Grantham JA, et al. The retrograde technique for recanalization of chronic total occlusions: a step-by-step approach. JACC Cardiovasc Interv 2012;5:1-11. [Crossref] [PubMed]
- Han Y, Jing Q, Su Y, et al. The revascularization of RCA-CTO procedure. Asvide 2019;6:213. Available online: http://www.asvide.com/watch/32898
Cite this article as: Han Y, Jing Q, Su Y, Wang W. Successful revascularization of right coronary artery chronic total occlusion by the antegrade and retrograde approaches. AME Case Rep 2019;3:30.


