Radiation induced rectal cancer: a case report with review of the literature
Introduction
Colorectal cancer is the third diagnosed cancer in men and the second in women, with an estimated of 1.4 million cases and 693,900 deaths occurring in 2012. The risk of colorectal cancer is higher within men (1).
The main risk factors for colorectal cancer are age above 50 years, inflammatory bowel diseases, personal or family history of adenoma or colorectal cancer, genetic predisposition, excessive consumption of red meat, alcohol, smoking and obesity (2-11).
The role of pelvic radiotherapy in the induction of rectal cancer has been found in several cases of rectal cancers after pelvic irradiation for a primary cancer other than the rectum. In this article we describe the case of a patient who developed a rectal adenocarcinoma, 18 years after being irradiated for squamous cell carcinoma of the cervix.
Case presentation
Our patient is a 73 years old woman, not a cigarette smoker or alcoholic, followed in our hospital since 1999 for squamous cell carcinoma of the cervix and presented 18 years after rectal bleeding and a rectal syndrome that revealed a well differentiated adenocarcinoma of the rectum. She has no history of inflammatory bowel disease or polyp and no history of cancer in the family. The squamous cell carcinoma of the cervix was according to the FIGO classification classified as stage IIB. The patient received external radiotherapy at a dose of 46 Gray with brachytherapy supplement at the dose of 26 Gray followed by a radical hysterectomy extended to both parameters with pelvic lymph node dissection. Subacute inflammatory rearrangements associated with a few perennial cervical tumor residues, with an inflammatory vaginal collar with some non-perennial tumor residues, were found in the histological study.
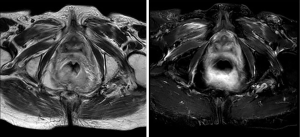
The patient was put under surveillance. She remained in good control until August 2017 when she presented rectal bleeding with a rectal syndrome. A hemi-circumferential budding process located at 2 cm from the anal margin was found in the digital rectal examination. She weighed 60 kilograms, heighted 160 cm with a body mass index of 23.4. The rest of the physical examination was normal. The colonoscopy found an ulcerative budding process 2 cm from the anal margin without other abnormalities in the rest of the colon. A well differentiated adenocarcinoma was diagnosed in the histological study. The chest-abdomen-pelvis CT scan performed as part of the extension assessment showed a thickening of the lower rectum without secondary locations. Pelvic magnetic resonance imaging showed thickening of the lower rectum without mesorectal adenomegaly (Figure 1). Tumor markers CEA and CA 19-9 were normal (CEA at 2.5 nanograms per milliliter and CA19-9 at 12 international units per milliliter).
The patient’s file was discussed at a multidisciplinary consultation meeting and a surgery was indicated at first. She had benefited from abdominoperineal resection with permanent left iliac fossa end colostomy.
The pathological analysis of the resection specimen showed a well differentiated adenocarcinoma infiltrating the rectal wall to perirectal fat with clearance of 1 millimeter, lymphovascular and perineural invasion were absent, proximal and distal margins were free, no ganglion was positive on 3 ganglions removed. The tumor was classified pT3N0M0 according to the TNM classification.
No adjuvant treatment was received and the patient is currently under control, with no evidence of local or metastatic recurrence with a follow-up of 8 months.
Discussion
Cervical cancer is the second most common cancer in women after breast cancer (1). Radiotherapy occupies an angular place in the treatment of cervical cancer (12,13). Improvement in overall survival reveals long term complications in long term survivors. Among these complications is the possibility of a radio induced secondary cancer. Cahan et al. are among the first to introduce the notion of secondary radio induced cancer, in 1948, by describing a series of 11 cases and they proposed criteria defining the radio induced tumors which are: tumor located in the initial field of irradiation, having a different histology from the initial lesion which appears after a relatively long delay of more than 5 years after irradiation (14).
PubMed research has found reports of isolated cases of rectal cancer after cervical cancer radiotherapy (15-18). In these publications, the delay between secondary cancer and radiotherapy was variable but relatively long ranging from 12 to 46 years.
Rodriguez and al conducted a study based on SEER data on 64,507 patients with cervical cancer, more than half of whom (52.6%) received pelvic radiotherapy (19). The authors showed in this study that there is no significant difference in the risk of occurrence of colorectal cancer in patients who received radiotherapy versus patients who did not receive it, although the risk becomes significant after 8 years for colon cancer and after 15 years for rectal cancer. Based on these results, the authors propose early detection of colorectal cancer in patients who have not reached the age of 50, 8 years after cervical cancer irradiation. These results were confirmed by a second study that showed a significant difference in the occurrence of rectal cancer in patients receiving radiation therapy on cervical cancer versus those who did not receive it (482 vs. 108) (20).
Our patient presented an adenocarcinoma of the rectum 18 years after being irradiated for her squamous cell carcinoma of the cervix. This delay is consistent with what it has been found in the literature. She presents criteria to diagnose a radio induced cancer proposed by Cahan et al. Also, she presents only one risk factor for colorectal cancer: age over 50 years. Considering all these elements, it is thought that this is a radio induced cancer more than a non-radio induced cancer.
Conclusions
We report a rare case of rectal adenocarcinoma in previously irradiated patient for cervical cancer. Available literature data currently show that rectal cancer may be a late complication of radiotherapy. We must think about considering this entity in monitoring protocols for long term survival patients who have been irradiated for cervical cancer.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this Case Report and any accompanying images.
References
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. [Crossref] [PubMed]
- Faivre J, Bouvier AM, Bonithon-Kopp C. Epidemiology and screening of colorectal cancer. Best Pract Res Clin Gastroenterol 2002;16:187-99. [Crossref] [PubMed]
- Butterworth AS, Higgins JP, Pharoah P. Relative and absolute risk of colorectal cancer for individuals with a family history: a meta-analysis. Eur J Cancer 2006;42:216-27. [Crossref] [PubMed]
- Johns LE, Houlston RS. A systematic review and meta-analysis of familial colorectal cancer risk. Am J Gastroenterol 2001;96:2992-3003. [Crossref] [PubMed]
- Green RJ, Metlay JP, Propert K, et al. Surveillance for second primary colorectal cancer after adjuvant chemotherapy: an analysis of Intergroup 0089. Ann Intern Med 2002;136:261-9. [Crossref] [PubMed]
- Botteri E, Iodice S, Bagnardi V, et al. Smoking and colorectal cancer: a meta-analysis. JAMA 2008;300:2765-78. [Crossref] [PubMed]
- Wu AH, Paganini-Hill A, Ross RK, et al. Alcohol, physical activity and other risk factors for colorectal cancer: a prospective study. Br J Cancer 1987;55:687-94. [Crossref] [PubMed]
- Must A, Jacques PF, Dallal GE, et al. Long-term morbidity and mortality of overweight adolescents. A follow-up of the Harvard Growth Study of 1922 to 1935. N Engl J Med 1992;327:1350-5. [Crossref] [PubMed]
- Rustgi AK. The genetics of hereditary colon cancer. Genes Dev 2007;21:2525-38. [Crossref] [PubMed]
- Gillen CD, Andrews HA, Prior P, et al. Crohn's disease and colorectal cancer. Gut 1994;35:651-5. [Crossref] [PubMed]
- Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut 2001;48:526-35. [Crossref] [PubMed]
- Marth C, Landoni F, Mahner S, et al. Cervical cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2017;28:iv72-83. [Crossref] [PubMed]
- Somashekhar SP, Ashwin KR. Management of Early Stage Cervical Cancer. Rev Recent Clin Trials 2015;10:302-8. [Crossref] [PubMed]
- Cahan WG, Woodard HQ, Higinbotham NL, et al. Sarcoma arising in irradiated bone: report of eleven cases. 1948. Cancer 1998;82:8-34. [Crossref] [PubMed]
- Tsunoda A, Shibusawa M, Kawamura M, et al. Colorectal cancer after pelvic irradiation: case reports. Anticancer Res 1997;17:729-32. [PubMed]
- Kimura T, Iwagaki H, Hizuta A, et al. Colorectal cancer after irradiation for cervical cancer--case reports. Anticancer Res 1995;15:557-8. [PubMed]
- Shirouzu K, Isomoto H, Morodomi T, et al. A case of radiation-induced rectal cancer developing after a long-term follow-up. Jpn J Clin Oncol 1994;24:294-8. [PubMed]
- Hareyama M, Okubo O, Oouchi A, et al. A case of carcinoma of the rectum after radiotherapy for carcinoma of the cervix. Radiat Med 1989;7:197-200. [PubMed]
- Rodriguez AM, Kuo YF, Goodwin JS. Risk of colorectal cancer among long-term cervical cancer survivors. Med Oncol 2014;31:943. [Crossref] [PubMed]
- Chaturvedi AK, Engels EA, Gilbert ES. Second cancers among 104,760 survivors of cervical cancer: evaluation of long-term risk. J Natl Cancer Inst 2007;99:1634-43. [Crossref] [PubMed]
Cite this article as: Dahiri M, Salmi N, Ahallat A, El Bahaoui N, Belkouchi O, Souadka A, Majbar A, Benkabbou A, Bougtab A, Mohsine R. Radiation induced rectal cancer: a case report with review of the literature. AME Case Rep 2018;2:31.


